Bacteria Present in the __________ Can Be Phagocytosed by Paneth Cells.
11.3E: Phagocytosis
- Page ID
- 3280
Learning Objectives
- Briefly describe the role of the following as they chronicle to phagocytosis:
- inflammation
- lymph nodules
- lymph nodes
- spleen
- Depict the following steps in phagocytosis:
- activation
- chemotaxis
- zipper (both unenhanced and enhanced)
- ingestion
- destruction
- State what happens when either phagocytes are overwhelmed with microbes or they adhere to cells to large to be phagocytosed.
- Describe what causes most of the tissue destruction seen during microbial infections.
- Compare the oxygen-dependent and oxygen-independent killing systems of neutrophils and macrophages.
- Briefly depict the role of autophagy in removing intracellular microbes.
Phagocytic cells include neutrophils, eosinophils, monocytes, macrophages, dendritic cells, and B-lymphocytes. Phagocytosis is the primary method used by the body to remove free microorganisms in the blood and tissue fluids. The body's phagocytic cells are able to come across these microorganisms in a variety of ways
Infection or tissue injury stimulates mast cells, basophils, and other cells to release vasodilators to initiate the inflammatory response. Vasodilation results in increased capillary permeability, enabling phagocytic white blood cells such as neutrophils, monocytes, and eosinophils - as well every bit other leukocytes - to enter the tissue around the injured site. The leukocytes are then chemotactically attracted to the area of infection. In other words, inflammation allows phagocytes to enter the tissue and go to the site of infection. Neutrophils are the commencement to appear and are after replaced by macrophage.
Lymph nodules are unencapsulated masses of lymphoid tissue containing fixed macrophages and ever irresolute populations of B-lymphocytes and T-lymphocytes. They are located in the respiratory tract, the liver, and the gastrointestinal tract and are collectively referred to equally mucosa-associated lymphoid tissue or MALT. Examples include the adenoids and tonsils in the respiratory tract and the Peyer'south patches on the small intestines. Organisms entering these systems can be phagocytosed past fixed macrophages and dendritic cells and presented to B-lymphocytes and T-lymphocytes (including T4 and T8-Lymphocytes ) to initiate adaptive allowed responses.
Tissue fluid picks up microbes then enters the lymph vessels every bit lymph. Lymph vessels deport the lymph to regional lymph nodes (Figure \(\PageIndex{1}\)). Lymph nodes contain many reticular fibers that back up fixed macrophages and dendritic cells besides every bit ever irresolute populations of circulating B-lymphocytes and T-lymphocytes. Microbes picked up by the lymph vessels are filtered out and phagocytosed in the lymph nodes by these fixed macrophages and dendritic cells and presented to the circulating B-lymphocytes and T-lymphocytes to initiate adaptive immune responses. The lymph eventually enters the circulatory system at the centre to maintain the fluid book of the circulation.
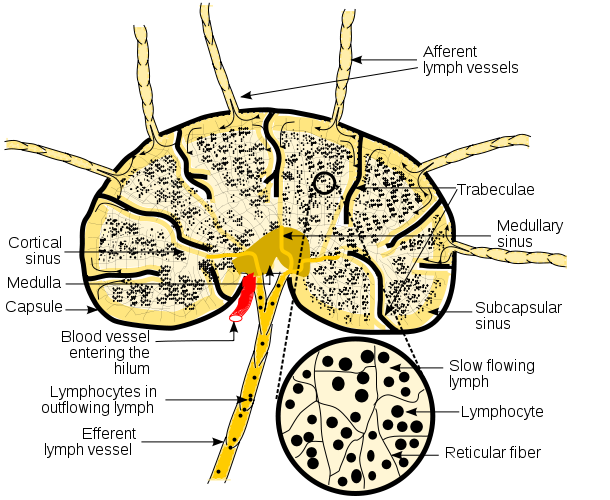
Figure \(\PageIndex{1}\): Diagram of a Lymph Node. Schematic diagram of a lymph node showing catamenia of lymph through lymph sinuses. Prototype used wtih permission (Public Domain; KC Panchal).
In addition, Langerhans' cells (immature dendritic cells) are located throughout the epithelium of the skin, the respiratory tract, and the alimentary canal where in their young form they are attached by long cytoplasmic processes. Upon capturing antigens through pinocytosis and phagocytosis and becoming activated by proinflammatory cytokines, the dendritic cells detach from the epithelium, enter lymph vessels, and are carried to regional lymph nodes. By the time they enter the lymph nodes, they accept matured and are at present able to present antigen to the e'er changing populations of naive T-lymphocytes located in the cortex of the lymph nodes.
The spleen contains many reticular fibers that back up fixed macrophages and dendritic cells, too every bit ever changing populations of circulating B-lymphocytes and T-lymphocytes. Blood carries microorganisms to the spleen where they are filtered out and phagocytosed by the stock-still macrophages and dendritic cells and presented to the circulating B-lymphocytes and T-lymphocytes to initiate adaptive immune responses. There are too specialized macrophages and dendritic cells located in the brain (microglia), lungs (alveolar macrophages), liver (Kupffer cells), kidneys (mesangial cells), bones (osteoclasts), and the gastrointestinal tract (peritoneal macrophages).
The Steps Involved in Phagocytosis
In that location are a number of distinct steps involved in phagocytosis:
Step one: Activation of the Phagocyte
Resting phagocytes are activated by inflammatory mediators such as bacterial products (bacterial proteins, capsules, LPS, peptidoglycan, teichoic acids, etc.), complement proteins, inflammatory cytokines, and prostaglandins. As a effect, the circulating phagocytes produce surface glycoprotein receptors that increase their ability to attach to the inner surface of capillary walls, enabling them to squeeze out of the capillary and be attracted to the site of infection.
In addition, they produce endocytic pattern-recognition receptors that recognize and demark to pathogen-associated molecular patterns or PAMPs - components of common microbial molecules such as peptidoglycan, teichoic acids, lipopolysaccharide, and mannose-rich glycans that are not found in human cells - to attach the microbe to the phagocyte for what is chosen unenhanced attachment (discussed beneath). They also showroom increased metabolic and microbicidal activity by increasing their production of ATPs, lysosomal enzymes, lethal oxidants, etc.
Step 2: Chemotaxis of Phagocytes (for wandering macrophages, neutrophils, and eosinophils)
Chemotaxis is the movement of phagocytes toward an increasing concentration of some attractant such equally bacterial factors (bacterial proteins, capsules, LPS, peptidoglycan, teichoic acids, etc.), complement proteins (C5a), chemokines (chemotactic cytokines such as interleukin-8 secreted by diverse cells), fibrin split products, kinins, and phospholipids released by injured host cells.
Movie showing chemotaxis past neutrophil. Chemotaxis of Neutrophils. © From Intimate Strangers: Unseen Life on World. Created by Mondo Media. Peter Bakery, Executive Producer. Licensed for use, ASM MicrobeLibrary.
Y'all Tube blitheness summarizing phagocytosis by a macrophage.
You Tube flick illustrating chemotaxis.
Some microbes, such as the influenza A viruses, Mycobacterium tuberculosis, blood invasive strains of Neisseria gonorrhoeae, and Bordetella pertussis take been shown to cake chemotaxis.
Pace 3: Attachment of the Phagocyte to the Microbe or Jail cell
Attachment of microorganisms is necessary for ingestion. Attachment may be unenhanced or enhanced.
a. Unenhanced attachment: Unenhanced attachment is the innate recognition of pathogen-associated molecular patterns or PAMPs - components of mutual molecules such as peptidoglycan, teichoic acids, lipopolysaccharide, mannans, and glucans mutual in microbial cell walls only not found on human cells - past means of endocytic blueprint-recognition receptors, such as scavenger receptors and mannose receptors, on the surface of the phagocytes (Figure \(\PageIndex{2}\)).
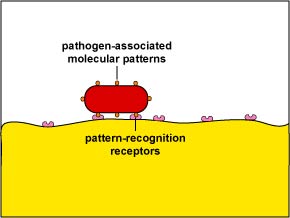
Effigy \(\PageIndex{two}\): Unenhanced Attachment of Bacteria to Phagocytes. Glycoprotein molecules known as blueprint-recognition receptors are found on the surface of phagocytes. They are so named considering they recognize and demark to pathogen-associated molecular patterns - components of common molecules such equally peptidoglycan, teichoic acids, lipopolysaccharide, mannans, and glucans - found in many microorganisms.
b. Enhanced attachment: Enhanced attachment is the zipper of microbes to phagocytes by fashion of an antibody molecule called IgG, the complement proteins C3b and C4b produced during the complement pathways (Figure \(\PageIndex{3}\)), and acute phase proteins such every bit mannose-binding lectin (MBL) and C-reactive protein (CRP). Molecules such as IgG, C3b, and mannose-bounden lectin (MBL) that promote enhanced attachment are chosen opsonins and the procedure is likewise known as opsonization. Enhanced zipper is much more specific and efficient than unenhanced.
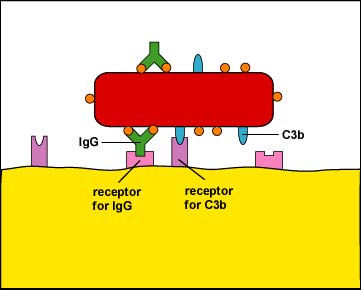
Effigy \(\PageIndex{3}\): Enhanced Attachment of Bacteria to Phagocytes. One of the functions of certain antibiotic molecules known equally IgG is to stick antigens such as bacterial proteins and polysaccharides to phagocytes. The "tips" of the antibody, the Fab portion, accept a shape that fits epitopes, portions of an antigen with a complementary shape. The "stalk" of the antibody is called the Fc portion and is able to bind to Fc receptors on phagocytes. Also, when body defense pathways known as the complement pathways are activated, i of the benign defense force proteins made is chosen C3b. C3b binds by one end to bacterial surface proteins and past the other stop to C3b receptors on phagocytes. The IgG and C3b are also known as opsonins and the process of enhanced attachment is besides called opsonization.
c. Extracellular trapping with NETs: In response to sure pathogen associated molecular patterns such as LPS, and certain cytokines such as IL-eight, neutrophils release DNA and antimicrobial granular proteins. These neutrophil extracellular traps (NETs) bind to bacteria, prevent them from spreading, and impale them with antimicrobial proteins (meet Effigy \(\PageIndex{fifteen}\) and Effigy \(\PageIndex{16}\)).
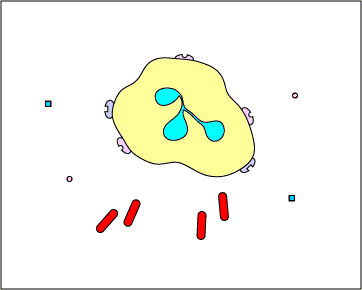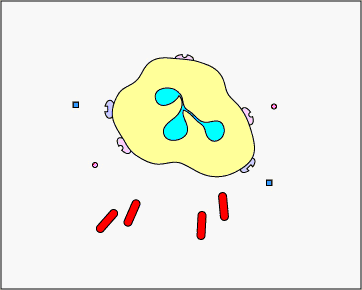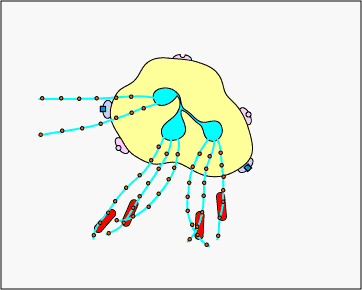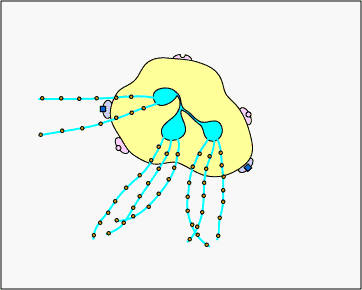
Neutrophil NETS Trapping and Killing Bacteria. In response to certain pathogen associated molecular patterns such as LPS, and sure cytokines such equally IL-8, neutrophils release DNA and antimicrobial granular proteins. These neutrophil extracellular traps (NETs) bind to bacteria, prevent them from spreading, and kill them with antimicrobial proteins such as histones and elastins. 1 hypothesis, shown in this animation, proposes that the NETs are produced by living neutrophils in response to bacteria. Alternately, NETs may exist released as a effect of necrotic cell expiry of neutrophils.
Some microorganisms are more resistant to phagocytic attachment.
a. Capsules can resist unenhanced zipper by preventing the endocytic pattern recognition receptors on phagocytes from recognizing the bacterial prison cell wall components and mannose-containing carbohydrates (see Figure \(\PageIndex{fourteen}\)). Streptococcus. pneumonia activates the classical complement pathway, but resists C3b opsonization, and complement causes further inflammation in the lungs.
c. Other capsules, rich in sialic acrid, a common component of host cell glycoprotein, accept an affinity for serum protein H, a complement regulatory protein that leads to the deposition of the opsonin C3b past gene I and the germination of C3 convertase. (Serum protein H is what ordinarily leads to the deposition of whatsoever C3b that binds to host glycoproteins and so that nosotros don't stick our own phagocytes to our own cells with C3b.)
d. Some capsules simply cover the C3b that does bind to the bacterial surface and prevent the C3b receptor on phagocytes from making contact with the C3b (come across Figure \(\PageIndex{3}\)). This is seen with the capsule of Streptococcus pneumoniae.
e. Neisseria meningitidis has a capsule equanimous of sialic acrid while Streptococcus pyogenes (grouping A beta streptococci) has a sheathing made of hyaluronic acid. Both of these polysaccharides closely resemble carbohydrates found in human tissue polysaccharides and because they are not recognized as strange by the lymphocytes that acquit out the immune responses, antibodies are non made against these capsules. Likewise, some bacteria are able to coat themselves with host proteins such as fibronectin, lactoferrin, or transferrin and in this mode avoid antibodies.
f. An outer membrane molecule of Neisseria gonorrhoeae called Protein II and the M-protein of Streptococcus pyogenes permit these bacteria to exist more resistant to phagocytic engulfment. The M-protein of S. pyogenes, for example, binds factor H of the complement pathway and this results in the degradation of the opsonin C3b by gene I and the germination of C3 convertase. Southward. pyogenes also produces a protease that cleaves the complement protein C5a.
m. Staphylococcus aureus produces protein A while Streptococcus pyogenes produces protein G. Both of these proteins bind to the Fc portion of antibodies (run across Effigy \(\PageIndex{4}\)) and in this way the leaner become coated with antibodies in a fashion that does not result in opsonization (encounter Figure \(\PageIndex{five}\)).
Footstep 4: Ingestion of the Microbe or Cell by the Phagocyte
Following attachment, polymerization and so depolymerization of actin filaments send pseudopods out to engulf the microbe (meet Figure \(\PageIndex{6}\)) and place it in an endocytic vesicle called a phagosome (run into Figure \(\PageIndex{7}\)).
During this process, an electron pump brings protons (H+) into the phagosome. This lowers the pH within the phagosome to three.5 - 4.0 so that when a lysosome fuses with the phagosome, the pH is correct for the acid hydrolases to effectively pause down cellular proteins. The acidification also releases defensins, cathelicidin, and bacterial permeability inducing poly peptide (BPI), peptides and enzymes that can kill microbes, from a matrix and enabling their activation.
Scanning electron micrographs of a macrophage with pseudopods and a macrophage phagocytozing E. coli on a blood vessel ; courtesy of Dennis Kunkel'due south Microscopy .
Intracellular microbes, such as viruses and bacteria that invade host cells, tin also be engulfed in one case they enter the cytosol of the cell by a process chosen autophagy. A membrane-bound compartment called an autophagosome grows around the microbe and the surrounding cytosol and afterwards delivers information technology to lysosomes for devastation (see Figure \(\PageIndex{17}\)). (This process is also used past eukaryotic cells to engulf and degrade unnecessary or dysfunctional cellular components such as damaged organelles.)
Some microorganisms are more than resistant to phagocytic ingestion
a. Pathogenic Yersinia, such equally the one that causes plague, contact phagocytes and, by ways of a type Three secretion organisation, deliver proteins which depolymerize the actin microfilaments needed for phagocytic engulfment into the phagocytes (run across Figure \(\PageIndex{viii}\)). Another Yersinia protein degrades C3b and C5a.
b. Some leaner, like Mycobacterium tuberculosis, Salmonella, and Listeria monocytogenes can block autophagy.
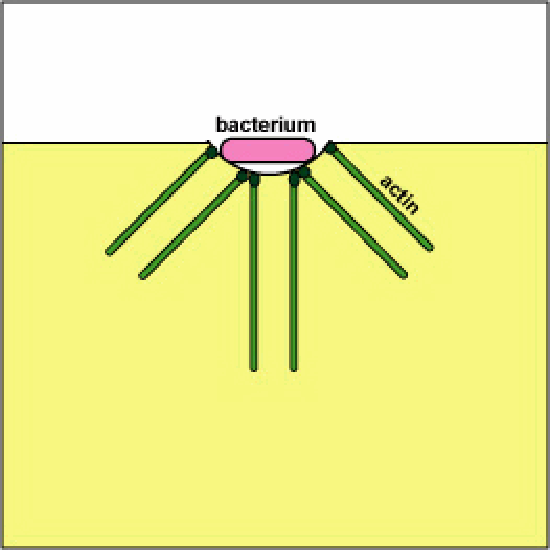
Blocking Phagosome Formation past Depolymerizing Actin. Molecules of some leaner, through a type Three secretion arrangement, evangelize proteins which depolymerize the phagocyte's actin microfilaments used for phagocytic engulfment.
Step 5: Destruction of the Microbe or Prison cell
Phagocytes incorporate membranous sacs chosen lysosomes produced by the Golgi apparatus that incorporate various digestive enzymes, microbicidal chemicals, and toxic oxygen radicals. The lysosomes travel along microtubules inside the phagocyte and fuse with the phagosomes containing the ingested microbes and the microbes are destroyed (see Figure \(\PageIndex{9}\)).
To view an electron micrograph of a phagolysosome, see the Spider web folio for the Academy of Illinois College of Medicine.
You Tube animation summarizing phagocytosis by a macrophage.
Some bacteria are more resistant to phagocytic destruction once engulfed.
a. Some leaner, such as Legionella pneumophilia and Mycobacterium species, cause the phagocytic prison cell to place them into an endocytic vacuole via a pathway that decreases their exposure to toxic oxygen compounds.
b. Some bacteria, such equally Salmonella, are more resistant to toxic forms of oxygen and to defensins (toxic peptides that impale bacteria).
c. Some bacteria, such as Shigella flexneri and the spotted fever Rickettsia, escape from the phagosome into the cytoplasm prior to the phagosome fusing with a lysosome (see Figure \(\PageIndex{10}\)).
d. Neisseria gonorrhoeae produces Por protein (protein I) that prevents phagosomes from fusing with lysosomes enabling the bacteria to survive inside phagocytes.
e. Some leaner, such every bit species of Salmonella, Mycobacterium, Legionella, and Chlamydia, block the vesicular transport machinery that enables the phagosome to fuse with the lysosome.
f. Some leaner, such every bit pathogenic Mycobacterium and Legionella pneumophilia, prevent the acidification of the phagosome which is needed for effective killing of microbes by lysosomal enzymes. (Unremarkably after the phagosome forms, the contents become acidified because the lysosomal enzymes used for killing function much more finer at an acidic pH.)
g. The carotenoid pigments that give Staphylococcus aureus its golden color and grouping B streptococci (GBS) its orange tint shield the bacteria from the toxic oxidants that neutrophils utilise to kill leaner.
h. Jail cell wall lipids of Mycobacterium tuberculosis, such every bit lipoarabinomannan, arrest the maturation of phagosomes preventing delivery of the bacteria to lysosomes.
i. Some bacteria are able to impale phagocytes. Bacteria such equally Staphylococcus aureus and Streptococcus pyogenes produce the exotoxin leukocidin which damages the cytoplasmic membrane of the phagocyte. On the other hand, bacteria, such every bit Shigella and Salmonella, induce macrophage apoptosis, a programmed prison cell expiry.
If the the infection site contains very large numbers of microorganisms and high levels of inflammatory cytokines and chemokines are beingness produced in response to PAMPs, the phagocyte will empty the contents of its lysosomes past a process called degranulation in order to kill the microorganisms or cell extracellularly.
These released lysosomal contents, however, besides kill surrounding host cells and tissue. Nigh tissue devastation associated with infections is a result of this process (see Effigy \(\PageIndex{11}\)).
The phagocyte will also empty the contents of its lysosomes for extracellular killing if the cell to which the phagocyte adheres is too large to be engulfed (see Figure \(\PageIndex{12}\) and Figure \(\PageIndex{xiii}\)).
There are 2 killing systems in neutrophils and macrophages: the oxygen-dependent system and the oxygen-independent system.
1. The oxygen-dependent organization: production of reactive oxygen species (ROS)
The cytoplasmic membrane of phagocytes contains the enzyme oxidase which converts oxygen into superoxide anion (O 2- ). This can combine with water past manner of the enzyme dismutase to form hydrogen peroxide (H 2 O 2 ) and hydroxyl (OH) radicals.
In the case of neutrophils, but not macrophages, the hydrogen peroxide can and so combine with chloride (Cl 2- ) ions by the activeness of the enzyme myeloperoxidase (MPO) to form hypochlorous acid (HOCL), and singlet oxygen.
In macrophages, nitric oxide (NO) tin can combine with hydrogen peroxide to course peroxynitrite radicals. (In improver to ROS and NO, macrophages secrete inflammatory cytokines such as TNF-blastoff, IL-ane, IL-eight, and IL-12 to promote an inflammatory response.)
These compounds are very microbicidal because they are powerful oxidizing agents which oxidize well-nigh of the chemical groups found in proteins, enzymes, carbohydrates, DNA, and lipids. Lipid oxidation can break down cytoplasmic membranes. Collectively, these oxidizing free radicals are called reactive oxygen species (ROS).
Oxidase too acts as an electron pump that brings protons (H+) into the phagosome. This lowers the pH within the phagosome and then that when lysosomes fuse with the phagosome, the pH is correct for the acid hydrolases, like elastase, to effectively break downwards cellular proteins.
In addition to phagocytes using this oxygen-dependant system to impale microbes intracellularly, neutrophils also routinely release these oxidizing agents, too equally acrid hydrolases, for the purpose of killing microbes extracellularly. These agents, even so, also current of air upwardly killing the neutrophils themselves also equally some surrounding torso cells and tissues as mentioned above.
2. The oxygen-independent system
Some lysosomes contain defensins ), cationic peptides that alter cytoplasmic membranes; lysozyme, an enzyme that breaks down peptidoglycan, lactoferrin, a protein that deprives leaner of needed iron; cathepsin One thousand, a protease that causes harm to microbial membranes; elastase, a protease that kills many types of bacteria; cathelicidins, proteins that upon cleavage are directly toxic to a multifariousness of microorganisms; bactericidal permeability inducing protein (BPI ), proteins used by neutrophils to impale certain leaner past damaging their membranes; collagenase ; and diverse other digestive enzymes that showroom antimicrobial activity by breaking downwards proteins, RNA, phosphate compounds, lipids, and carbohydrates.
Exercise: Call up-Pair-Share Questions
- Streptococcus pyogenes has a capsule made of hyaluronic acid, a polysaccharide also found on human cells.
Draw what significance this has in the bacterium resisting phagocytosis and why.
- Streptococcus pyogenes produces a protein called protein G that binds to the Fc portion of homo IgG.
Draw what significance this has in the bacterium resisting phagocytosis and why.
- Many bacteria produces capsules that comprehend their cell wall.
Draw what significance this has in the bacterium resisting phagocytosis and why.
- People born with chronic granulomatious illness accept neutrophils that lack the enzyme oxidase in their cytoplasmic membrane.
Depict what significance this has in the bacterium resisting phagocytosis and why.
Summary
Phagocytosis is the chief method used by the trunk to remove costless microorganisms in the blood and tissue fluids. An inflammatory response to injury and/or infection allows phagocytes to leave the bloodstream, enter the tissue, and go to the site of infection or injury. Microorganisms inbound lymph nodules found in the respiratory, gastrointestinal, and genitourinary tract can be phagocytosed past fixed macrophages and dendritic cells and presented to B-lymphocytes and T-lymphocytes to initiate adaptive immune responses.Tissue fluid picks up microbes in the tissue, enters the lymph vessels as lymph, and carries the microbes to regional lymph nodes where they are filtered out and phagocytosed by stock-still macrophages and dendritic cells and presented to the circulating B-lymphocytes and T-lymphocytes to initiate adaptive immune responses.
Dendritic cells located throughout the epithelium of the pare, the respiratory tract, and the gastrointestinal tract phagocytize microbes, enter lymph vessels, and carry the microbes to regional lymph nodes where the dendritic cells present antigens associated with the microbes to the ever irresolute populations of naive T-lymphocytes.Claret carries microorganisms to the spleen where they are filtered out and phagocytosed past fixed macrophages and dendritic cells and presented to the circulating B-lymphocytes and T-lymphocytes to initiate adaptive immune responses. There are also specialized macrophages and dendritic cells located in the encephalon (microglia), lungs (alveolar macrophages), liver (Kupffer cells), kidneys (mesangial cells), bones (osteoclasts), and the alimentary canal (peritoneal macrophages.
- Resting phagocytes are activated by inflammatory mediators and produce surface receptors that increase their ability to adhere to the inner surface of capillary walls enabling them to squeeze out of the capillary and enter the tissue, a process called diapedesis.
- Activation likewise enables phagocytes to produce endocytic design-recognition receptors that recognize and demark to microbial PAMPs in gild to attach the microbe to the phagocyte, likewise every bit to exhibit increased metabolic and microbicidal activity.
- Phagocytes then use chemotaxis to move towards an increasing concentration of some attractant such as bacterial factors or defense molecules.
- Attachment of phagocytes to the microbes or cells can be through unenhanced attachment or enhanced attachment.
- Unenhanced attachment is the recognition of pathogen-associated molecular patterns or PAMPs by endocytic pattern-recognition receptors on the surface of the phagocytes.
- Enhanced attachment, or opsonization, is the zipper of microbes to phagocytes by style of an antibody molecule called IgG, the complement proteins C3b and C4b, and acute stage proteins such as mannose-binding lectin (MBL) and C-reactive protein (CRP).
- Following zipper, polymerization and so depolymerization of actin filaments send pseudopods out to engulf the microbe and identify it in an endocytic vesicle called a phagosome.
- During this process, an electron pump brings protons (H+) into the phagosome to lowers the pH within the phagosome to a pH that is correct for the acid hydrolases to effectively suspension down cellular proteins.
- Phagocytes contain bleary sacs called lysosomes that contain various digestive enzymes, microbicidal chemicals, and toxic oxygen radicals. The lysosomes fuse with the phagosomes containing the ingested microbes and the microbes are destroyed.
- If the infection site contains very large numbers of microorganisms and high levels of inflammatory cytokines and chemokines are being produced in response to PAMPs, the phagocyte volition empty the contents of its lysosomes in order to kill the microorganisms or prison cell extracellularly.
- Lysosomal contents released during extracellular killing likewise kill surrounding host cells and tissue. Most tissue devastation associated with infections is a result of extracellular killing by phagocytes.
Contributors and Attributions
-
Dr. Gary Kaiser (COMMUNITY College OF BALTIMORE County, CATONSVILLE CAMPUS)
Source: https://bio.libretexts.org/Bookshelves/Microbiology/Book%3A_Microbiology_(Kaiser)/Unit_5%3A_Innate_Immunity/11.4%3A_Early_Induced_Innate_Immunity/11.3E%3A_Phagocytosis

0 Response to "Bacteria Present in the __________ Can Be Phagocytosed by Paneth Cells."
Post a Comment